-
Publish Your Research/Review Articles in our High Quality Journal for just USD $99*+Taxes( *T&C Apply)
Offer Ends On
Judy R Wilson* and Abu B Yilla
Corresponding Author: Judy R Wilson, Department of Kinesiology, University of Texas at Arlington, Arlington, Texas 76019, USA
Received: February 12, 2022 ; Revised: March 07, 2022 ; Accepted: March 09, 2022 ; Available Online: April 30, 2022
Citation: Wilson JR & Yilla AB. (2022) The Immediate Effects of Maximal Aerobic and Anaerobic Exercise on Mood. J Psychol Psychiatr Res, 1(1): 1-9.
Copyrights: ©2022 Wilson JR & Yilla AB. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Views & Citations
Likes & Shares
Introduction: Numerous studies have used the Profile of Mood States (POMS) to assess exercise effects on mood and have suggested that low to moderate exercise is preferred due to the resulting positive mood changes such as vigor and exhilaration. The purpose of this study was to determine maximal exercise effects on the mood states of college-aged males and females.
Methods: The short form of the POMS was administered for the three exercise protocols, Bruce and Ramp for aerobic exercise, and the Wingate Anaerobic Test for anaerobic exercise. Participants (n=55) were oriented with an initial visit while the order of the other sessions was randomized. The POMS was given pre-exercise, post-exercise and 20 min into recovery.
Results: The results indicated that the Total Mood Disturbance scores were significantly higher (almost double) following the maximal aerobic exercise (13.51 ± 8.1) than the maximal anaerobic exercise (7.40 ± 8.77; p< 0.001).
Discussion: The primary finding of this study was that the maximal aerobic test (Bruce Protocol) elicited greater Total Mood Disturbances than the Wingate Anaerobic Test. However, following the 20 min recovery, all measures returned to baseline.
Keywords: Profile of Mood States, Physical activity, Intensity, Health
INTRODUCTION
It has been known for some time that a physically active lifestyle results in both physical and psychological health benefits [1-3]. To increase the outreach of this information the Healthy People Initiative was launched. It began with the Surgeon General's report [4] and new iterations of the Healthy People Initiative have followed every decade since 1980. Progress has been made in reducing the major causes of death such as heart disease and cancer. Reductions in risk factors like smoking, high blood pressure and high cholesterol, have also been made [5]. In the snapshot report for Healthy People 2020 there was a positive result with the number of adults meeting aerobic and physical activity and muscle-strengthening objectives increasing significantly from 18.2% in 2008 to 24.0% in 2018 [6]. The challenge remains as to how to interest the rest of the adult population to participate in activities that will improve both physical and mental health.
Maximal aerobic exercise represents a level of exertion that is generally left to research laboratories to measure or to clinicians for diagnosis of coronary artery disease and exercise prescription [7]. Maximal oxygen uptake (VO2max) is an important physiological variable correlated with exercise endurance and is also a predictor of cardiovascular mortality [8], while maximal anaerobic power is often overlooked. But peak muscle power and muscle endurance are fitness components that are also important. For some daily activities, as well as athletic events, it is essential to develop high-intensity power that is available instantaneously. The development of the Wingate Anaerobic Test (WAnT) has made it possible to assess muscle power, muscle endurance and fatigability [9] and is independent of oxygen utilization [10]. While both exercise tests represent 'maximal intensity exercise' [11]. one represents the maximal capacity of the muscles to use oxygen (VO2max) and the other represents the capacity to use the stores of energy (phosphagens) [10] in the skeletal muscles (WAnT). With differing physiological demands [12] would these different types of exercise result in differences in mood as assessed by the short form of the Profile of Mood States (POMS)? [13].
Studies that have focused on the effects of exercise over time (chronic) have found a positive association between mood and the amount of time spent in exercise [14,15]. However, our approach was designed to evaluate mood changes in response to a single (acute) bout of exercise. There is evidence to support that an acute bout of aerobic exercise is sufficient to change mood that can last up to 30 min [16]. Cycling for 15 min increased positive affect in young and old adults [17] and resulted in reduced sensitivity to anxiety after walking or running [18].
The frequency of exercise and the duration of each exercise bout are part of the "exercise prescription." Intensity is also an important part of the exercise prescription and has been considered the most extensively studied, moderating factor [19]. However, the effect of intensity on mood changes has not been consistently observed. Moderate-intensity exercise was found to be superior to either low- or high- intensity aerobic exercise for reducing negative affect and increasing positive affect [20]. Moderate- and high-intensity exercise have improved depression levels [21] and improved mood after high-intensity running [22]. Finding the proper intensity for physical activity is important to maintaining adherence and decreasing the risk of dropout [23]. Self-selected levels of intensity may be the most beneficial choice for adherence and positive mood changes [24].
High intensity exercise is not generally recommended because of the potential to decrease adherence to exercise programs [25]. It is encouraging then, that research has shown improvements in mood do result from low-intensity activities such as walking [26] up through exercise intensities that reach ventilatory and/or lactate threshold [27]. Mood responses above this threshold are almost always negative compared to the positive mood states for low- to moderate-intensities [23]. Additional research against the benefits of high intensity exercise indicated that affect was least positive during exercise above the lactate threshold than during self-selected and below the lactate-threshold conditions [28]. What is also of interest is that after high intensity exercise is completed, mood states are usually positive, regardless of intensity [23].
Maximal exercise provides a relatively standard exercise load that allows for comparisons across studies. It also provides evidence to support or refute the idea that highly strenuous exercise has a negative impact on mood [29].
Pronk, Crouse and Rohack [30] used the abbreviated form of the POMS to determine the effects of a maximal exercise test on the mood of middle-aged females (45 ± 2.5 years). Maximal exercise was found to increase fatigue and decrease tension and vigor as measured 5 min post exercise. Using subscales from the POMS, it was determined that tension and anxiety were reduced following maximal exercise in active men, but not among the sedentary group [31]. In other research, the POMS ratings of 11 collegiate cyclists to 8 college students were compared following three exercise tests. No differences were found between the two groups pre- to post- maximal exercise. Both the cyclists and the students showed increases in depression, fatigue, and confusion along with decreases in vigor [32]. Due to conflicting results during dose-response studies, it had been concluded that fitness levels were not a factor [33]. However, the impact of fitness level on exercise-induced changes in mood may be seen when higher intensities are introduced. Differences in mood responses to exercise between high- and low-fit individuals was demonstrated when the intensity level was highly demanding such as during a maximal exercise test [34]. Steptoe, Kearsley, and Walters [31] found a significantly greater decrease in tension-anxiety in trained individuals following maximal exercise than in sedentary individuals.
The standard Bruce protocol has been widely used since its development in 1963 [35]. However, this protocol, that is performed on a treadmill, includes sudden changes in speed and elevation that cause a high oxygen consumption with each 3 min stage. This often leads to the individual stopping early before reaching their age-predicted maximum heart rate. A ramped protocol uses more modest changes in speed and elevation that is better tolerated [36]. Thus, it was decided to include two different maximal aerobic exercise tests to determine if this resulted in differences in mood.
The effect of anaerobic exercise on mental indices has not been studied as extensively as the effect on aerobic exercise. A comparison between the two types of exercise (aerobic and anaerobic) were evaluated in previous research. A ten-week training program resulted in improvement in the mean scores of anxiety, stress, and happiness for both groups. However, the group who trained anaerobically had significant improvements in stress levels and happiness over the aerobically-trained group [37]. Two different types of anaerobic training programs also had a positive impact on state anxiety in college athletes [38].
The relationship between mood and anaerobic power was evaluated to determine whether time of day had any effect. Four Wingate anaerobic power tests were conducted at various times throughout a single day. The authors found that greater levels of fatigue, as indicated by the POMS scale, were associated with reduced performance and dependent on the time of day [39].
The Wingate test has also been utilized to determine effects of rest in underperforming elite competitors [40]. Olympic athletes were allowed to rest for 3-5 weeks following which aerobic and anaerobic exercise tests were read ministered. There was a significant reduction in fatigue and mood profile scores and a significant increase in vigor following the Wingate test when compared to the mood scores prior to the rest.
The purposes of the study were to determine the immediate effects of maximal aerobic and anaerobic exercise on mood states in college students as well as any differences in mood between maximal aerobic exercise protocols.
METHODS
Sample
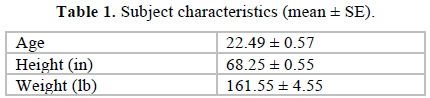
Sixty-two college students participated in this study and 55 (88.7%) completed all four visits to the Exercise Science Laboratory on the university campus. Flyers advertising the study were posted around the University campus asking for volunteers. The order that participants took the tests was randomized by the drawing of lots (labeled table tennis balls). Participants ranged in age from 18 to 36 years and included 27 males and 28 females. The subject characteristics are presented in Table 1. The mean maximum oxygen consumption (VO2max) for the 55 subjects obtained for the Bruce protocol was 39.91 ± 1.0 ml/kg/min and for the Ramp, 41.58 ± 1.1 ml/kg/min, and did not differ significantly between the exercise modalities. Similarly, there was no significant difference between the heart rate maximums for the Bruce (184.04±2.0 bpm), and the Ramp (181.07 ± 1.5 bpm). The Wingate Anaerobic test elicited mean maximal peak responses of 608.7 ± 22.6 W.
Measures
The short form of the Profile of Mood States (POMS) [41] was utilized to assess mood. This instrument is a tool used to assess mood state in both active and inactive populations. The POMS is a 37-item inventory designed to gauge the participant’s global mood state. The items are divided into six factors (tension, depression, anger, vigor, fatigue, and confusion) and rated on a 5-point Likert scale. A seventh scale, total mood disturbance, can be derived by calculation from the other scales. Adequate internal consistency and test-retest reliability have been ascertained [13].
Procedures
Those who expressed an interest in the study were asked to sign a written informed consent form approved by the Institutional Review Board. The initial visit to the laboratory was an orientation to collect data on height, weight, and percent body fat. Each participant was also acquainted with the mouthpiece and headgear and experienced walking on the treadmill through the first stage of the Bruce protocol. The short version of the POMS was administered immediately before the exercise protocol began (pre) and again immediately after the subject stepped off the treadmill or stopped pedaling on the bicycle ergometer (post). At 20 min into recovery, the POMS was readministered (recovery). Participants were asked to respond to the questionnaire regarding their feelings “right now.”
The following three visits were randomly assigned. Two visits involved maximal aerobic tests that included the Bruce protocol and a Ramp protocol. The Bruce protocol starts at a speed of 1.7 mph at a 10% grade. The workload increases every 3 min by increasing the elevation by 2% and the speed by 0.8 to 0.9 mph. While the Bruce protocol is commonly used, some individuals have difficulty with the awkward pace at various levels. Consequently, a Ramp protocol was added to determine if varying the process of achieving maximal capacity would affect mood. The Ramp protocol increased elevation 0.5% every 12s (2.5% per min) and increased speed by ½ mile/h every 3 min (beginning at 3.5 miles/h). The Wingate Anaerobic Power test [30] consisted of 30s of high speed pedaling (100 rpms). The resistance was set at 0.075 kg/kg body mass and was applied within 3s after the subject overcame the initial inertia.
Statistical analysis
For results from the POMS, seven 2 x 2 repeated measures ANOVAs (Exercise [Bruce, Ramp, Wingate] by Time [Pre, Post, Recovery]) were employed. The scales for each analysis were the 7 scales of the POMS; Tension, Depression, Anger, Vigor, Fatigue, Confusion, and Total Mood Disturbance. Due to the multiple analyses, a Bonferroni adjustment was made to the alpha level of .05 resulting in a test alpha of 0.007 (05/7). When the ANOVA revealed significant effects, a Tukey post-hoc analysis was performed. When the ANOVA revealed a significant interaction between exercise and time one-way ANOVAs were performed and tested at alpha = 0.05. If data sets failed Mauchly’s test of sphericity for the repeated measures designs a Greenhouse-Geiser adjustment was employed. All data were analyzed utilizing SPSS 26.
RESULTS
There were no significant differences between the mood effects of the Bruce and the Ramp tests. Therefore, for clarity, only the results of the tests between the Bruce and the Wingate tests are presented here. Table 2 provides the maximal values for all exercise tests.
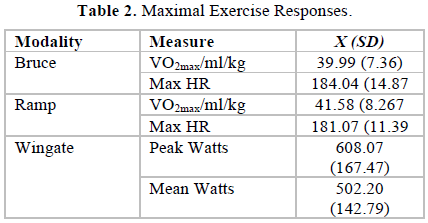
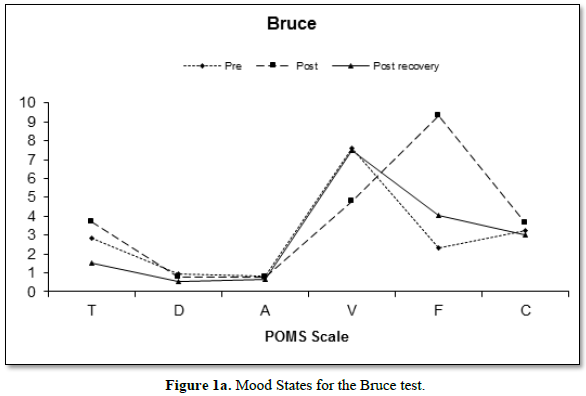
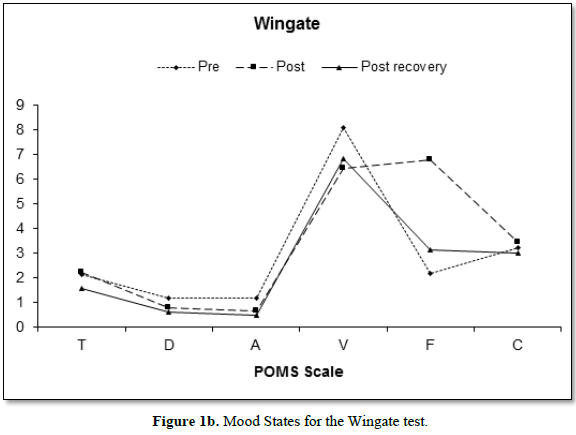
Figure 1 depicts the mean changes in mood states of both the Bruce (Figure 1a) and Wingate (Figure 1b) protocols. The reduction in vigor between the pre- and post- scores for both the Bruce and the Wingate are of note, as is the increase in fatigue.
Table 3. presents the significant effects of the 2 x 2 ANOVAs by the POMS scales. There were no significant (p > 0.007) differences on the anger scale.
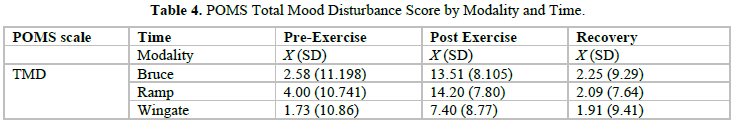
Table 4 provides the information regarding the Total Mood Disturbance scores obtained before, after and into recovery for each modality.
Interactions
As can be seen from Table 3, there were significant interactions on the scales for total mood disturbance (F[4, 216] = 9.04, p< 0.001), tension (F[4, 216] = 9.02, p< 0.001), vigor (adjusted F[2.89, 155.88] = 4.53, p = 0.005), and fatigue (F [4, 216] = 7.87, p< 0.001). Follow-up one-way ANOVAs on exercise and time were performed for each of these scales. The interactions for these four scales are presented in Figure 2
Total Mood Disturbance
The one-way ANOVAs on the Bruce test for the total mood disturbance scale yielded a significant main effect between the pre, post and recovery scores (F[2, 108] = 41.86, p< 0.001) with significant differences between pre (2.58 ± 11.2) and post scores (13.51 ±8.1) and post (13.51 ± 8.1) and recovery scores (2.25 ± 9.3). Total mood disturbance (Table 4) was therefore immediately elevated by exercise and then returned to pre-exercise levels. There was a similar pattern on the Wingate test (F [2, 108] = 14.57, p<0.001) with the pre (1.73 ± 10.9) and post (7.40 ± 8.8) scores differing significantly, as well as the post (7.40 ± 8.8) and recovery s (1.91 ± 9.4) scores. The only significant difference between exercise protocols was that of the post scores on the Bruce and the Wingate tests (F [2, 108] = 27.98, p<0.001), with the Bruce (13.5 ± 8.1) eliciting a higher mood disturbance score than the Wingate (7.40 ± 1.6).


Tension
With the examination of tension, there was a significant difference between all three conditions on the Bruce test (adjusted F [1.75, 94.50] = 20.13, p<0.001; pre-2.82± 2.9; post3.71 ±2.4; recovery 1.51 ± 1.8). The Wingate test elicited a significant main effect F (2, 108) = 3.60, p =0.03; with only the scores between post and recovery differing in the follow-up analysis (post 2.22 ± 2.17; recovery 1.56 ± 1.8). The Wingate test was seen to increase tension immediately post exercise and then reduce it in recovery, but not to levels significantly below that of the pre-exercise state.
Vigor
The Bruce protocol elicited a significant main effect for vigor F (2, 108) = 11.13, p<0.001; with the scores between the pre (7.58 ± 4.6) and the post (4.78 ± 3.6) and the post (4.78 ± 3.6) and recovery (7.47 ± 4.5) differing in the follow-up analysis. The Wingate test also elicited a significant main effect F (2, 108) = 40.31, p =0.005 with the pre (8.1 ± 3.8) scores differing from both the post (6.44 ± 3.5) and the recovery measures (6.82 ± 3.9). The Bruce and the Wingate tests elicited a significantly different main effect for time F (2, 108) = 8.94, p<0.001, with the tests differing on the post measures only (B:4.78 ± 3.6; W: 6.44 ±3.5).
Fatigue
Both the Bruce and the Wingate tests elicited significant main effects for fatigue (Bruce, F [2, 108] = 91.50, p<0.001; Wingate, F [2, 108] = 53.62, p<0.001). Both protocols elicited significant differences on pre (B:2.33 ±2.4; W: 2.16 ± 2.8) and post (B:9.35 ± 4.0; W: 6.76 ± 4.2), pre and recovery (B: 4.02 ± 3.4; W: 3.13 ± 3.4), and post and recovery scores. Fatigue peaked immediately post exercise and remained above pre-exercise levels through the recovery phase. The post exercise measure was the only one for which the exercises significantly differed when directly compared F (2, 108) = 22.83, p
Time
There were no significant interactions, or exercise effects, on the depression and confusion scales. On the significant main effect for time on the depression scale (adjusted F[1.70, 91.46] = 10.71, p =0.002), a post-hoc analysis identified a significant reduction in depression between pre and recovery scores (X = 0.98, X = 0.52, respectively), and post and recovery scores (X = 0.79, X = 0.52). On the confusion scale a main effect for time was identified (F [2, 108] = 8.96, p<0.001). Post-hoc analysis identified significant differences between pre- and post- scores (X = 3.30, X = 3.42) and post and recovery scores (X = 3.42, X = 3.21). Therefore, confusion increased immediately post exercise and then returned to pre-exercise levels during recovery.
DISCUSSION
Fifty-five college-aged men and women completed the two maximal aerobic exercise tests on the treadmill using different protocols (Bruce, Ramp) and a maximal anaerobic test on a bicycle ergometer (Wingate). In this study, subjects were recruited from the general student population and on average, were low to moderately fit. A standard maximal aerobic test allowed for a consistent end point across subjects as well as providing data to identify any differences in mood between the two types of maximal exercise (aerobic vs anaerobic).
The findings of significant increases in the Total Mood Disturbance (TMD) scores pre- to post exercise across all three exercise tests agree with Pronk et al. (1995) who found an increase in the TMD scores pre- to post exercise [42]. However, an unanticipated finding was that the increase in TMD scores for the Wingate was half the TMD scores resulting from the treadmill tests (Figure 2).
All three of these exercise tests are considered “maximal” in effort and intensity. Yet, the most obvious difference is the time each exercise test takes to complete. The WAnT is set at a standard time of 30s, although some may choose 60s [9]. A maximal exercise test such as the Bruce or Ramp will vary in the time it takes the participant to reach maximal capacity which was average (10 ± 2.1 min) in this study. The differences in time may be thought to elicit differences in physiological plasma levels of norepinephrine which has been suggested to have an effect on mood [43]. However, evidence in the literature suggests that these levels can be similar, 4.25 nmol/L for maximal oxygen consumption testing [44] and 3.75 nmol/L for the WAnT [45].
A thermogenic hypothesis has also been suggested as a mechanism for improved mood states associated with physical activity [46]. This question was addressed and found that elevated body temperature may not be necessary for exercise-related anxiety reduction to occur [47]. Again, the differences in time of exercise would indicate a difference in the increase in both core and muscle temperature between maximal aerobic and maximal anaerobic exercise.
It is interesting to theorize as to why differences were found between these two types of exercise. The goal for people who are new to exercising should be an emphasis on the psychological responses rather than the physiological responses. High intensity exercise produces less mood improvement [48] while exercising at a preferred level of intensity has more of a desirable effect on mood [49]. Exercise duration has also been evaluated to determine its influence on mood changes [50]. The American College of Sports Medicine has recommended 10 min exercise sessions to accumulate the 30 min per day considered to benefit health [51]. Further study has indicated that short duration exercise (15 min) does result in desirable mood changes [52].
The general finding of this study was that the maximal exercise test, using the Bruce Protocol, elicited stronger and more lasting effects on mood than the Wingate, except for vigor. However, the considerable difference in time of exercise between the aerobic and anaerobic tests must be considered a major contributing factor. The average time for the tests on the treadmill was approximately 10 min compared to 30 s on the bicycle ergometer. A repeat of the maximal exercise test utilizing a cycle ergometer would keep the modalities consistent and may yield different effects on changes in mood.
Issues to address in future research would include the relationship between the Ratings of Perceived Exertion (RPE) and mood changes. Do those with higher RPE values at VO2max also report greater disturbances in mood? Also, the effects of maximal exercise on mood disturbance may vary with age. The mean age of the sample in this study was 22 ± 0.57 years. The responses to such high intensity exercise may vary among middle-aged and older adults and with fitness level. In addition, the mood responses following maximal exercise in untrained subjects may differ following a period of aerobic and/or anaerobic training.
CONCLUSION
The purpose of this study was to evaluate the effects of maximal types of exercise on mood. While the results offer interesting findings as to the mood changes elicited by each type of exercise, maximal exercise should not be how sedentary individuals should be recruited to an exercise program. While a ‘stress’ test is often ordered by a physician to diagnose cardiac disease, it might discourage the novice individual. The findings that support the benefits of regular exercise in reducing risks of cardiovascular disease have led to efforts to reverse the decline in participation in physical activity in America. Other research has explored the psychological benefits that result from regular exercise. Despite a wide variation in research design, the conclusions drawn from most research is that regular physical activity is beneficial to both physiological and psychological health. Whatever the type, intensity or duration, the best exercise is the one that will result in consistent participation.
No Files Found
Share Your Publication :